1994 Nobel Prize for the discovery of G proteins in signal transduction
- The Natural Philosopher
- Oct 10, 2020
- 3 min read
By Marlena Tyldesley
In 1994, the Nobel Prize in Physiology or Medicine was awarded to both Alfred G. Gilman and Martin Rodbell for their discovery of G proteins and their role in signal transduction in cells (The Nobel Prize in Physiology or Medicine 1994, n.d.).
The story:
Alfred Gilman, who conducted research on Leukemia cells, had found that these cells were not reacting to outside stimulation by hormones. Gilman and his team found that there was one absent protein in the cells responsible for this inability to take in signals from outside of them. The transplantation of a protein from normal, functional cells into these leukemia cells restored their ability to interpret and transmit signals. Gilman would eventually name this protein the G-protein. “G”, because the protein binds to a molecule called guanosine triphosphate (GTP). Gilman once said that these proteins are involved in “everything from sex in yeast to cognition in humans” (Alfred Goodman Gilman, M.D., Ph.D., n.d.).
Martin Rodbell, in his own research, had discovered the framework of a cell communication pathway, but it had a missing piece - Gilman’s G protein. Rodbell knew that GTP was crucial to his pathway, and Gilman’s discovery of the G protein gave the mechanism by which GTP was transduced inside the cell, completing Rodbell’s signal transduction pathway (Grimes, 2015). As such, they were both awarded the Nobel Prize in 1994 (The Nobel Prize in Physiology or Medicine 1994, n.d.).
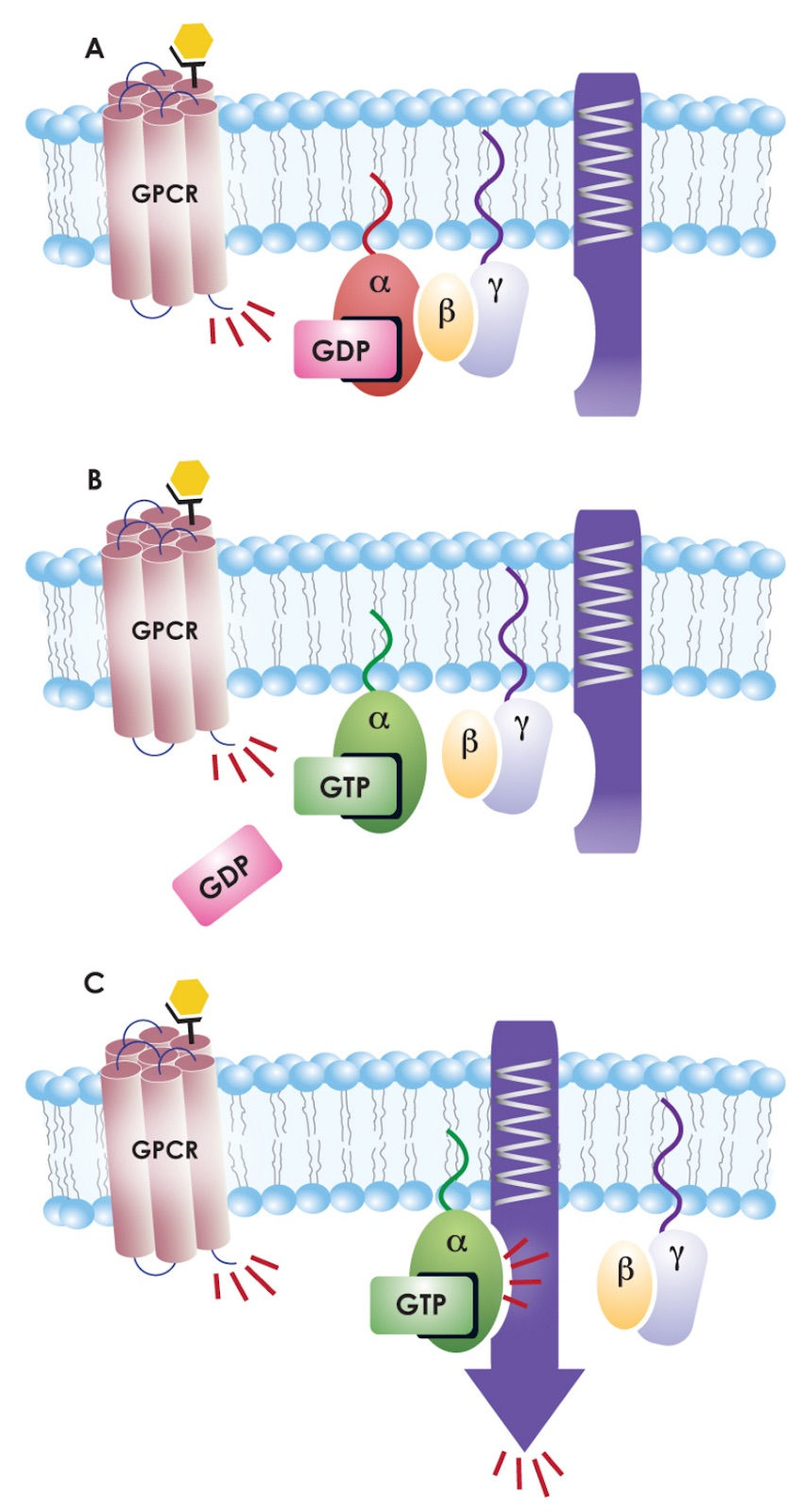
G Proteins and Their Coupled Receptors, 2012
G proteins are made up of three subunits: alpha, beta and gamma (shown above in green, yellow and light purple, respectively). The activating molecule for G proteins, and one of the molecules Rodbell identified as crucial during his research, is GTP, or guanosine triphosphate. In their inactive state, G-proteins are associated with GDP (guanosine diphosphate), shown in bright pink, instead of GTP. When a molecule binds the GPCR, though, the now active receptor converts GDP to GTP. GTP can then activate the G protein, which will go on to activate any of a number of pathways (G Proteins and Their Coupled Receptors, 2012).
Gilman’s and Rodbell’s joint discovery of the G-protein and its role in signal transduction is crucial in many fields. They have been used to develop medications that treat asthma and hypertension (Kamato et al., 2015) and different families of G protein may play roles in the heart’s ability to contract, in vision, in taste, and in the regulation of immune cells, to name only a very small selection (Syrovatkina et al., 2016). As someone who cannot stand to learn about the cell cycle year in and year out, when my introductory biology course finally moved on to G proteins, I breathed a sigh of relief. This article serves as my thank you to Alfred Gilman and Martin Rodbell for their discovery of a molecule that helped draw me to cellular biology, and that illustrates so perfectly the enormous impact of the things we cannot see.
Bibliography
Alfred Goodman Gilman, M.D., Ph.D. (n.d.). Yale Class of 1962. https://yale62.org/obituaries/gilman0716.html
G Proteins and Their Coupled Receptors. (2012). Cayman Chemical. https://www.caymanchem.com/news/g-proteins-and-their-coupled-receptors
Grimes, W. (2015, December 24). Dr. Alfred G. Gilman, Whose Work on Proteins Won Nobel Prize, Dies at 74. The New York Times. https://www.nytimes.com/2015/12/25/us/dr-alfred-g-gilman-whose-work-on-proteins-won-nobel-prize-dies-at-74.html
Kamato, D., Thach, L., Bernard, R., Chan, V., Zheng, W., Kaur, H., Brimble, M., Osman, N., & Little, P. (2015, March 24). Structure, function, pharmacology, and therapeutic potential of the G protein, Gα/q,11. Frontiers. https://www.frontiersin.org/articles/10.3389/fcvm.2015.00014/full
The Nobel Prize in Physiology or Medicine 1994. (n.d.). The Nobel Prize. https://www.nobelprize.org/prizes/medicine/1994/summary/
Syrovatkina, V., Alegre, K., Dey, R., & Huang, X.-Y. (2016, August 8). Regulation, Signaling and Physiological Functions of G-proteins. PMC. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5023507/



Comments